
Nijmegen, the Netherlands, 17 March, 2026 — MGI Tech Co., Ltd. (MGI), a company dedicated to developing tools and technologies that drive innovation in life sciences, today announced a major milestone in its collaboration with Predica Diagnostics. Predica’s HPV Profiler and CervicaDx assays, based on its proprietary Cirnaseq technology, have been successfully validated and deployed on MGI’s DNBSEQ sequencing platforms, delivering a dramatic reduction in turnaround time from two weeks to a single day.
The achievement marks the transition of the partnership from initial agreement less than two years ago to real-world impact. Predica has integrated MGI’s DNBSEQ platforms into both its internal R&D and commercial service projects, with multiple labs receiving results at speeds not possible on conventional platforms. The validation confirms that laboratories already running DNBSEQ systems can seamlessly incorporate Predica’s Cirnaseq-based diagnostics into their existing workflows.
A Complete, Scalable Solution for Cervical Cancer Screening
MGI’s E25 and G99 sequencers, combined with Predica’s Cirnaseq technology and Cirnalyzer software, deliver an affordable and scalable workflow for clinical labs, service providers, and national cervical cancer screening programs. The integrated solution offers:
Simultaneous high-resolution HPV genotyping and host-gene expression profiling
Improved specificity for cervical cancer risk assessment and HPV+ triage
Automated analysis and reporting through Predica’s Cirnalyzer software
Lower cost per sample
Faster R&D, Faster Results
In parallel, Predica’s team used the platform to run side-by-side comparisons of different library preparation conditions, rapidly identifying the optimal protocol (Figure 1) for the Cirnaseq platform. The kind of iterative optimization that used to take months can now be completed in days.
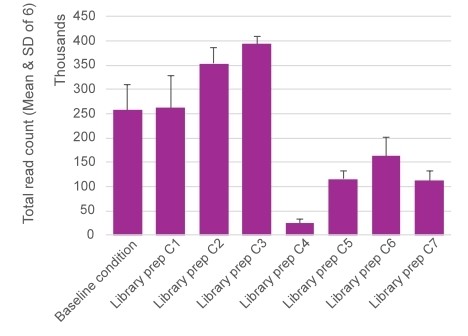
Figure 1: Optimization of library preparation on the MGI E25. Total Assigned Reads (mean ± SD) across tested conditions.
Enabling Access Through Technology
“By combining our Cirnaseq assays with MGI’s sequencing technology, we’re enabling labs to access deeper biological insights at a fraction of the cost,” said Dr. William Leenders, CSO of Predica Diagnostics. “This is part of our mission to improve women’s health.“
“By running Predica’s Cirnaseq assays on our DNBSEQ platforms, we’re proving that high-accuracy cancer diagnostics doesn’t have to come with a high price tag or slow turnaround. For the millions of women waiting on cervical cancer screening results, faster turnaround means less anxiety and faster treatment. That’s the impact we’re proud to enable.,” added Dr. Fang Chen, General Manager of Europe & Africa at MGI.
Looking ahead, MGI and Predica will jointly validate assays, develop native DNBSEQ library preparation kits, and prepare for IVDR submissions. Initial market focus will be on Europe and Africa, with global expansion to follow.
About Predica Diagnostics
Predica Diagnostics B.V. is a precision-oncology company developing targeted RNA sequencing tests for early cancer detection, risk stratification, and treatment guidance. Founded as a spin-off from Radboudumc, Predica aims to bring its Cirnaseq technology to clinical practice to reduce overdiagnosis and overtreatment and improve patient outcomes.
Learn more about Predica Diagnostics or contact the team at info@predicadx.com
Share






